SEOUL, May 11 (AJP) - A research team at Kookmin University has developed a new heterostructure catalyst that significantly improves the efficiency of hydrogen production in alkaline water electrolysis. The team, led by Professor Lee Chan-woo of the Department of Chemistry, identified the specific mechanism that allows the catalyst to accelerate the water-splitting process, the university said Monday.
The research focuses on Anion Exchange Membrane Water Electrolysis (AEMWE), a next-generation technology that operates in alkaline environments to reduce reliance on expensive precious metal catalysts and corrosion-resistant parts. While promising, these systems are often limited by a slow initial step where water molecules must be broken down to create hydrogen intermediates, leading to higher energy consumption and lower efficiency.
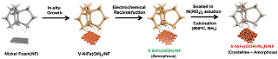
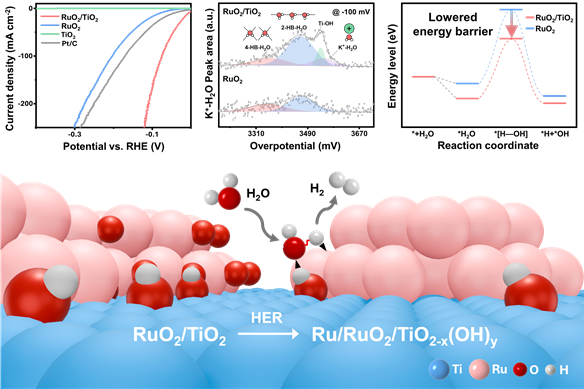
To address this bottleneck, the team engineered a catalyst by forming ruthenium oxide nanoparticles approximately 2 nanometers in size onto a 25-nanometer titania support. During the electrochemical process, the catalyst surface reconfigures itself into an active interface that attracts water molecules and lowers the energy barrier required to break their chemical bonds.
Test results showed that the new catalyst required an overpotential of only 6.6 millivolts to reach a standard current density. This performance significantly outperforms commercial platinum catalysts, which require 43 millivolts, and standalone ruthenium oxide, which requires 79 millivolts. The mass activity of the new material was found to be approximately 6.4 times higher than that of standard platinum-on-carbon catalysts.
The study utilized real-time spectroscopy and theoretical calculations to track water molecules and hydrogen intermediates during the actual reaction. This allowed the team to confirm that the titania interface directly facilitates the cutting of oxygen-hydrogen bonds. The researchers found that the ruthenium and titania work together, with the interface handling water activation while the ruthenium handles the formation of hydrogen molecules.
"This research is significant because we developed a high-activity catalyst while simultaneously observing how the interface activates water molecules under actual operating conditions," Professor Lee Chan-woo said. "Based on the principle of cooperative hydrogen formation at the ruthenium-titania interface, we can suggest design strategies for high-efficiency catalysts applicable to next-generation electrolysis systems."
The study was supported by the Ministry of Science and ICT and was published in the international journal Carbon Energy.
(Reference Information)
Journal/Source: Carbon Energy
Title: Ruthenium-Titania Interface-Mediated Water Activation for High Turnover Frequency in Alkaline Hydrogen Evolution
Link/DOI: https://doi.org/10.1002/cey2.518
Copyright ⓒ Aju Press All rights reserved.