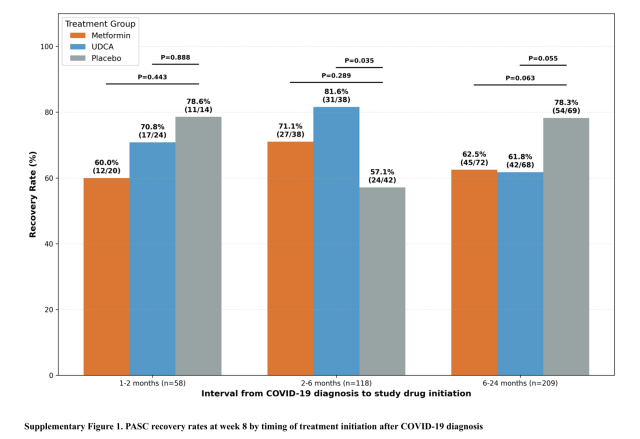
Daewoong Pharmaceutical said Monday that ursodeoxycholic acid, or UDCA, the main ingredient in its Urusa product, showed an improvement signal among long COVID patients who were 2 to 6 months past infection. The findings were posted online March 3, Eastern time, in the journal Annals of Internal Medicine.
In that 2-to-6-month group, 81.6% of patients receiving UDCA showed symptom improvement, significantly higher than 57.1% in the placebo group. By simple comparison, that was about 43% higher than placebo, the company said. No such improvement signal was seen in patients more than 6 months after infection.
The results suggest the importance of timing in long COVID treatment and, in an exploratory way, point to the potential for drug intervention earlier after infection.
Researchers also conducted immune analyses to examine inflammatory changes. Among patients whose symptoms improved, inflammation-related markers tended to decline, and the pattern was more pronounced in the 2-to-6-month group. The researchers said additional analysis is needed to determine whether the inflammation changes reflect a drug effect.

JW Pharmaceutical said Monday it held a symposium on its bowel-cleansing agent J-Cle at the 47th Spring Scientific Meeting of the Korean Society of Gastrointestinal Endoscopy, held March 8 at Lotte Hotel Seoul in the Sogong-dong area of Seoul.
Park Su-beom, a professor in the gastroenterology department at Pusan National University Yangsan Hospital, presented Phase 3 results for J-Cle and discussed how it can be used for preparation before colonoscopy.
J-Cle is a prescription drug taken to cleanse the colon before tests such as endoscopy and X-ray imaging. JW said the total dose is 20 tablets, and the film-coated tablets are designed to be easier to swallow. The company said powder or liquid bowel preps can cause adverse reactions such as nausea and headaches during dosing, increasing patient burden or reducing test accuracy, while tablet formulations can reduce adverse reactions and improve convenience.
J-Cle contains magnesium sulfate, potassium sulfate and sodium sulfate, which act as osmotic laxatives, along with simethicone to reduce foam in the colon and sodium picosulfate to promote bowel movement. Park said that, based on Phase 3 results, J-Cle showed bowel-cleansing efficacy comparable to the control group, with features related to safety and medication adherence.
Celltrion said Monday that its flagship treatment Remsima, whose active ingredient is infliximab, has demonstrated prescription performance in Asia with a dominant market share.
Citing IQVIA, Celltrion said Remsima held the top prescription share as of the third quarter of last year, with 93% in Singapore, 77% in Hong Kong, 73% in Thailand and 65% in Malaysia. Another autoimmune disease treatment, Yuflyma, whose active ingredient is adalimumab, rose to No. 2 in Singapore, surpassing the original product, the company said.
Celltrion said its local subsidiaries have increased bid-win rates in Asia’s tender-driven markets by emphasizing supply stability. In Thailand, the company said its local unit has built long-term cooperation with all university hospitals, which has helped win recognition for the quality competitiveness of key products including Remsima, Truxima and Herzuma. Celltrion said the three products are used exclusively at all university hospitals there. The company said it plans to expand its product portfolio in Asia based on the performance of its existing lineup.

Lotte Biologics said Monday it will participate in DCAT Week 2026, a global pharmaceutical and biotech business event, to be held March 23-26 in New York.
DCAT Week is an annual international networking event in New York where global drugmakers and contract development and manufacturing organizations, or CDMOs, seek collaboration opportunities.
Lotte Biologics said it will operate a dedicated meeting room at the Lotte New York Palace hotel during the event and pursue partnerships for its global CDMO business, including in North America. The company said it will highlight synergies between the quality competitiveness of its Syracuse Bio Campus in New York and the large-scale production capacity of the first plant at its Incheon Songdo Bio Campus, which is scheduled for completion in August.

Hugel said Monday it will work with the Korean Academy of Anti-Aging Dermatology, or KAAD, on an annual collaboration aimed at establishing global standards for botulinum toxin procedures and strengthening clinician education.
Hugel said it held the ‘Hugel X Korean Academy of Anti-Aging Dermatology Academic Collaboration Kickoff Meeting’ on March 6 at the Westin Josun Seoul Parnas, where the sides discussed developing treatment protocols for its botulinum toxin product Botulax, exported as Letybo, and plans for global webinars.
The two sides agreed to hold five global webinars every other month from April through December. Each session will include a lecture, live demonstration and Q&A. Hugel said real-time subtitles in English and Chinese will be provided for the convenience of clinicians worldwide.
* This article has been translated by AI.
Copyright ⓒ Aju Press All rights reserved.

